
Executive Summary
Consistent with standing precedents, the U.S. Court of Appeals for the Federal Circuit (the court) invalidated Seagen’s US10,808,039 patent (the ’039 patent) claims directed to antibody-drug conjugates (ADCs) comprising a subgenus of tetrapeptide linkers.1 The’039 patent claims generically cover Enhertu, an ADC, manufactured by Daiichi Sankyo (Daiichi), containing a tetrapeptide linker (GGFG) for treating cancer. The court invalidated the ’039 patent for failing to meet the written description and enablement requirements because the patent claims covered a subgenus of tetrapeptide linkers that was not sufficiently described in the priority application filed in 2004. The court also ruled that the ’039 patent was not entitled to the 2004 priority date, and Daiichi’s public disclosure of Enhertu in 2015 was intervening art and anticipated the ’039 patent. In view of this decision, patent owners should exercise heightened caution when drafting claims based solely on commercial interest (e.g., to cover a potential infringer’s product) rather than support in the priority documents.
Procedural History
In 2015, Daiichi publicly disclosed the structure of Enhertu, which includes its G/F tetrapeptide linker, GGFG. AstraZeneca joined Daiichi in commercializing Enhertu. In 2019, Seagen filed the patent application (later issued as the ’039 patent) directed to ADCs having tetrapeptide linkers having only glycine (G) or phenylalanine (F) and claimed priority to a Seagen patent application filed in 2004. After the ’039 patent issued in 2020, Seagen filed a patent infringement suit in district court against Daiichi, alleging that Enhertu infringed Seagen’s ’039 patent.2 Daiichi countered by arguing that the ’039 patent was invalid for lack of written description to support the claimed subgenus of glycine/phenylalanine (G/F) linkers, and for lacking an enabling disclosure that would allow a person of skill in the art to practice the full scope of the ADC claims without undue experimentation. A timeline of the key events prepared by the court is provided below.
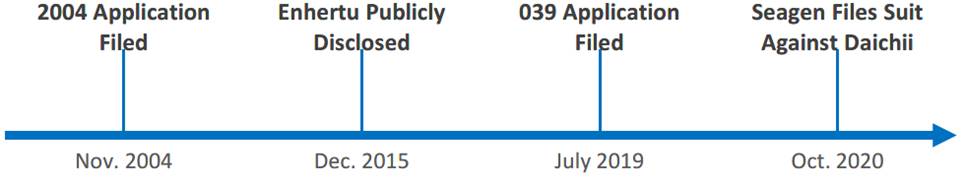
At the district court trial for patent infringement, the jury found that the ’039 patent satisfied both the written description and enablement requirements, and Daiichi’s Enhertu infringed the ’039 patent, awarding Seagen $41.82 million in damages. Daiichi appealed the verdict to the Federal Circuit (the court), which overturned the jury’s decision, finding the ’039 patent invalid for both lacking written description and enablement.3
The Court’s Decision: Written Description
The written description requirement reflects a basic premise of the patent system: an inventor may obtain a patent only if they disclose the invention to the public in sufficient enough detail that a person of ordinary skill in the art (POSA) would understand the inventor truly possessed the full scope of the claimed invention.4 For a claim covering a genus to satisfy the written description requirement, the specification must either: i) disclose a representative number of species falling within the scope of the claimed genus; or ii) disclose structural features common to all members of the genus (“common structural features test”).5
If the claim covers a previously undisclosed subgenus, the common structural features test requires that the patent disclosure provide sufficient “blaze marks”6 to direct a POSA to the claimed subgenus. The court has explained the “blaze marks” requirement with an analogy to a forest, where the forest represents a disclosure of a broad genus, and the trees represent particular subgenera or species within the genus. The court has indicated that there must be sufficient guidance (“blaze marks”) in the patent disclosure that leads a POSA to the claimed subgenera or species from the multitude of other possibilities covered by the genus. In other words, the court has found that a disclosure of a broad genus (forest) is insufficient to support a claimed subgenus or species (tree) without specific and descriptive detail (blaze marks or guidance) pointing one to the claimed invention.
Based on the blaze mark criteria, the court held that no reasonable jury could find that the ’039 patent had written description support from the 2004 priority application for the G/F tetrapeptide subgenus, because the 2004 priority application discloses a genus of 47 million tetrapeptide possibilities, and only three exemplified tetrapeptide sequences (GFLG, ALAL, GSVQ), none of which include a G/F-only linker. As such, the court held that the 2004 priority application does not support either i) a representative number of species falling within the scope of the claimed G/F tetrapeptide linker subgenus or ii) sufficient blaze marks to direct a POSA to the G/F tetrapeptide linker subgenus from the genus of 47 million tetrapeptide possibilities.
The court rejected Seagen’s argument that disclosure of the GFLG linker was a sufficient blaze mark to a G/F tetrapeptide linker. The court held that a POSA would not have made the logical leap to substitute the leucine (L) of the GFLG linker with phenylalanine (F) to arrive at the G/F tetrapeptide based on the disclosure of the 2004 priority application. The court pointed to Seagen inventors’ own testimonies to support that the inventors did not truly possess the claimed invention at the time of the 2004 patent application filing. Specifically, the court found that none of the inventors envisioned a G/F tetrapeptide in 2004, and the G/F tetrapeptide linker subgenus was only described for the first time when the ’039 patent was filed in 2019. Accordingly, the court ruled that the ’039 patent claims were not entitled to the 2004 priority date because the 2004 written description did not support the claimed subgenus. Instead, the court held that the ’039 patent claims were entitled to a priority date of 2019 (when the claims were first filed). Thus, under the court’s analysis, Enhertu, which was first publicly disclosed in December 2015, anticipated the ’039 patent.
The Court’s Decision: Enablement
The enablement requirement represents the quid pro quo of the patent system, which incentivizes innovation by way of awarding time-limited exclusivity rights to the patent owner while also disseminating the innovation for public benefit and use upon expiration of the patent. Specifically, in order for the patent owner to obtain the benefit of an exclusive right to prevent others from practicing a patented invention, the patent owner must enable those working in the field of the invention to “make and use” the full scope of the patented claims without undue experimentation.
In the present case, the scope of the ’039 patent covered ADCs having a drug moiety (D) connected to an antibody via a G/F tetrapeptide linker, where “the drug moiety is intracellularly cleaved in a patient from the antibody.” The court stated that to determine whether an ADC met this functional limitation would require undue experimentation due to the unpredictable nature of ADCs, and thus a POSA would be left to perform a “trial-and-error discover” to make and use the ADCs within the scope of the ’039 patent. As such, practicing the full scope of the claimed ADCs would be undue, and the patent was held to lack enablement.
Conclusion
The decision in the present case provides a reminder that the full scope of a patent’s claims must be sufficiently described in a manner that allows a POSA to practice it without undue experimentation. Patent drafters should be cognizant that a genus claim may be narrowed during patent prosecution to a subgenus, e.g., to avoid prior art, and thus should draft a robust disclosure with subgenera of varying scope in the patent, in particular subgenera that are supported with numerous exemplified species. This case illustrates the difficultly faced by those drafting undisclosed subgenera by picking and choosing substituents from a broad genus where there is no guidance in the patent as originally filed for making such choices. This case may affect a patent owner’s ability to later cover their products using early patent applications that do not specifically disclose such products. This case underscores the importance of careful patent drafting and regular communication between patent seekers and their patent counsel when filing patent applications.
For questions regarding patent strategies, please contact any member of Wilson Sonsini’s Patents and Innovations practice.
1Seagen Inc. v. Daiichi Sankyo Co., Ltd., 2023-2424 (Fed. Cir. Dec. 2, 2025).
2Seagen Inc. v. Daiichi Sankyo Co., Ltd., No. 2:2020cv00337.
3Separately, Daiichi was successful in instituting post grant review (PGR) against the ’039 patent after the jury verdict, arguing that the ’039 patent lacked written support for the G/F tetrapeptide and thus not was entitled to the 2004 priority date. SeeDaiichi Sankyo, Inc. v. Seagen Inc., No. PGR2021-00030 (P.T.A.B. Feb. 14, 2023).
4Regents of the Univ. of Minn. v. Gilead Scis., Inc., 61 F.4th 1350, 1355 (Fed. Cir. 2023).
5Ariad Pharms., Inc. v. Eli Lilly & Co., 598 F.3d 1336, 1350-52 (Fed. Cir. 2010).
Contributors
- Privacy Policy
- Terms of Use
- Accessibility





